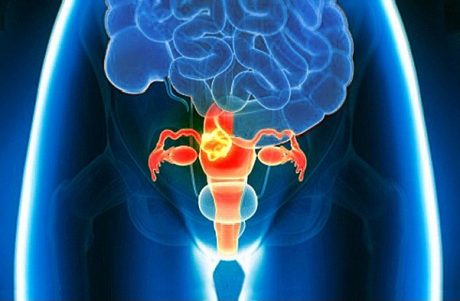
Multidisciplinary management, standards of care, optimal diagnostics, therapies and future perspectives in the care of GTN patients in sub-Saharan Africa.
-8th June 2020-1st July 2020-
Lead Faculty: Fredrick Chite Asirwa MD
Consultant Physician. Medical Oncologist & Hematologist
Executive Director/CEO: International Cancer Institute (ICI)
P.O. Box 8088, Code 30100 Eldoret, Kenya
Email: director@intercancer.com Website: www.intercancer.com
This educational activity is provided by the International Cancer Institute (ICI)
This activity is supported by an educational grant from Roche.
Faculty
1. Dr. Elly Odongo-Gynecologic Oncologist
2. Dr. Dawit Worku-Gynecologic Oncologist
3. Dr. Gregory Ganda– Gynecologic Oncologist
4. TBD– Mental Health Specialist
5. Dr. David Wata-Oncology Pharmacist
This course is accredited by:



Target Audience
The primary target audience for this course comprises the multidisciplinary clinical cancer care team members including, but not limited to medical, surgical, and radiation oncologists; fellows; residents; gastroenterologists; nephrologists; Obs/GYN; neurologists; pathologists; pulmonologists; radiologists; researchers; nurse practitioners and nurses; Midwives; pharmacists; PharmTechs; clinical officers; physician assistants; and other allied health care professionals.
Statement of Need
General internists and Primary Care Clinicians must maintain an extensive knowledge base on a wide variety of topics covering all body systems as well as common and uncommon disorders. International Cancer Institute’s Preceptorship Courses aims to leverage the expertise of its Faculty to help clinicians understand best practices in diagnosis and management of conditions encountered in the clinical setting in sub-Saharan Africa.
LEARNING OBJECTIVES
At the end of this educational activity, participants should be able to:
- Discuss the incidence and Epidemiology of GTN
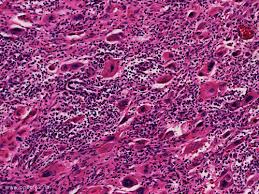
- Understand the clinical features and different types of GTN and their prognosis and molecular pathogenesis.
- Be familiar with the diagnostic and Imaging evaluation of GTN, including the role and limitations of computed tomography (CT), magnetic resonance imaging (MRI) and pelvic ultrasound and staging according to FIGO with providing (WHO) prognostic information for GTN
- Counsel patients on treatment for recurrent or persistent disease
- Understand the role of primary surgery in the management of complete and partial molar pregnancies
- Understand the management of GTN by FIGO stage, including the indications for single-agent versus multiple-agent chemotherapy, and the role of chemotherapy in the treatment of persistent or recurrent disease
- Understand the surveillance of GTN following treatment, including the importance of (and methods for) preventing subsequent pregnancy
Disclosure of Conflicts of Interest
International Cancer Institute (ICI) requires instructors, planners, managers and other individuals and
their spouse/life partner who are in a position to control the content of this activity to disclose any real or apparent conflict of interest they may have as related to the content of this activity. All identified conflicts of interest are thoroughly vetted by ICI for fair balance, scientific objectivity of studies mentioned in the materials or used as the basis for content, and appropriateness of patient care recommendations.
The faculty must report any financial relationships or contractual relationships they or their spouse/life partner have with commercial interests related to the content of this continuing education activity.
Disclaimer
Participants have an implied responsibility to use the newly acquired information to enhance patient outcomes and their own professional development. The information presented in this activity is not meant to serve as a guideline for patient management. Any procedures, medications, or other courses of diagnosis or treatment discussed in this activity should not be used by clinicians without evaluation of patient conditions and possible contraindications on dangers in use, review of any applicable manufacturer’s product information, and comparison with recommendations of other authorities.
Disclosure of Unlabeled Use.
This educational activity may contain discussion of published and/or investigational uses of agents that are not indicated by the FDA. The planners of this activity do not recommend the use of any agent outside of the labelled indications.
The opinions expressed in the educational activity are those of the faculty and do not necessarily represent the views of the planners. Please refer to the official prescribing information for each product for discussion of approved indications, contraindications, and warnings.
Method of Participation and Request for Credit
To receive credit for this activity, participants must review the activity information including learning objectives and faculty/planner disclosures and actively participate in the educational activity. Upon successfully completing the post-test with a score of 75% or better and the post-activity evaluation, your certificate will be made available immediately.
The Program
LECTURES
Orientation to the course (Chite) Monday 8th June 2020 4:00-4:30 PM EAT
- Incidence and Epidemiology of GTN (Dawit) Wednesday 10th June 2020 4:00-5:30 PM EAT
- Pathology, Staging and Risk Assessment of GTN (Elly) 15th June 2020 4:00-5:30 PM EAT
- Management of Low-Risk GTN (Ganda) 17th June 2020 4:00-5:30 PM EAT
- Management of High-Risk GTN (Dawit) 22nd June 2020 4:00-5:30 PM EAT
- Pharmacology of agents used in GTN management (Wata) 24th June 2020 4:00-5:00 PM EAT
Counselling patients on GTN treatments (TBC) 24th June 2020 5:00-5:30 PM EAT
- Management of recurrent and/or resistant GTN (Chite) 29th June 2020 4:00-5:30 PM EAT
- Summary of Recommendations (Standard of Care), Protocols and Surveillance (Dawit) 1st July 2020 4:00-5:30 PM EAT
All those interested in registration to this course, please send your information to info@intercancer.com
Acknowledgements
ICI Acknowledges that the funding for this preceptorship training has been proudly supported by Roche as part of ICI_Roche Shining Tower Project (STP).

Course Reviews
No Reviews found for this course.